TECHNICAL DATA SUMMARY
Parameter | Specification | Clinical Relevance |
Microsphere Size | 25 – 45 µm | Standard for facial/hand soft tissue augmentation. |
Composition | ~30% CaHA / 70% Gel | Matches volume/mass ratio of leading implants. |
Carrier Gel | Na-CMC + Glycerin | Replicates shear-thinning extrusion profile. |
Particle Morphology | Smooth, Spherical | Ensures smooth flow; mimics osteoconductive scaffold. |
Sterility | Non-Sterile | FOR EDUCATIONAL/LAB USE ONLY. |
THE COMPLETE TRAINING ECOSYSTEM
Material + Anatomy = Proficiency.
Advasaf has partnered with Ultrassist, the leader in high-fidelity medical simulators, to offer bundled training solutions. Combine the rheological precision of CAHA SIMULATE™ with anatomically correct silicone models for a "Turnkey" skills lab.
FEATURED BUNDLES
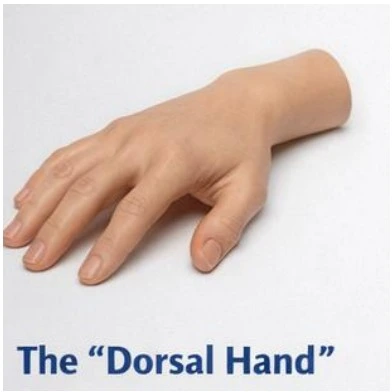
The "Dorsal Hand" Restoration Kit
Material: 5x Syringes of CAHA SIMULATE™ (1.5cc).
Model: Ultrassist Geriatric IV/Injection Hand.
Objective: Master the laminar plane separation between the dermis and superficial fascia. Visualize the masking of tendons and veins using the opaque CaHA paste.
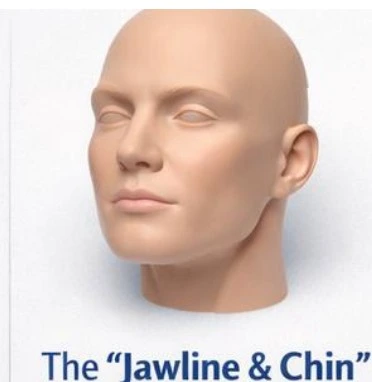
The "Jawline & Chin" Contouring Kit
Material: 10x Syringes of CAHA SIMULATE™ (1.5cc).
Model: Ultrassist Injection Head with bony landmarks.
Objective: Practice deep periosteal depot placement. Experience the high viscosity required to project tissue against muscle pressure.
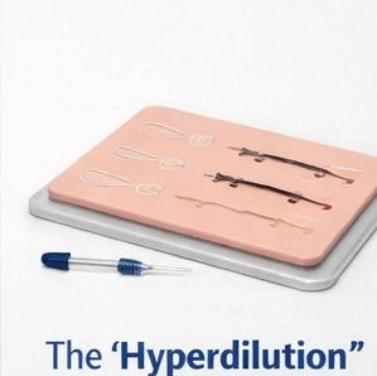
The "Hyperdilution" Workshop Kit
Material: 1x Bulk Jar of CAHA SIMULATE™ (50mL).
Model: Ultrassist Multi-Layer Suture/Injection Pad.
Objective: Train staff on mixing protocols (1:1 to 1:4 dilution with saline) for skin tightening and neocollagenesis stimulation.
ORDERING INFORMATION
CAHA SIMULATE™ is available in three formats to suit your facility's needs:
- Standard Training Pack: 10 x 1.5cc pre-filled syringes (Luer Lock).
- Laryngeal/Urology Pack: 10 x 0.8cc syringes (Precision Volume).
- Bulk Lab Jar: 50mL or 100mL jars (Ideal for R&D and large workshops).
REGULATORY NOTICE
CAHA SIMULATE™ is classified as a Non-Clinical Simulation Material.
NOT FOR HUMAN USE. This product is non-sterile and has not undergone viral inactivation or pyrogen testing required for human implantation.
Labeling: "For Training/Research Use Only."
Disposal: Contains biodegradable calcium phosphate; safe for standard waste disposal.